Most cancer patients are diagnosed and treated in community oncology practices. However, their participation in clinical trials is low. A common barrier to clinical trial enrollment in the community is the lack of research infrastructure and experienced research personnel.
Unique strategies to address the needs of community sites, especially those not affiliated with large cancer centers, are needed.
We describe how the Gulf South Clinical Trials Network, a minority/underserved National Cancer Institute Community Oncology Research Program community site, has developed Virtual Research Nurse program to provide virtual research support to urban and rural community oncology clinics to increase participation in cancer clinical trials.
The Gulf South Clinical Trials Network (CTN) is a minority/underserved National Cancer Institute Community Oncology Research Program community site that includes urban, suburban, and rural clinical sites throughout Louisiana and parts of Mississippi and Alabama.1
The Gulf South CTN has increased participation in clinical trials 10-fold, with more than 50% of participants being African American. However, most enrollments occur in major cities, while suburban and rural community practices struggle to enroll patients. Barriers to clinical trial participation in community oncology settings are multiple and complex. Most notable is the lack of research infrastructure and personnel to support clinical trials.2,3
Strategies to optimize research infrastructure and operations have been shown to improve accruals at large cancer centers.4-6 Since there is neither funding for nor the caseload to warrant full-time research staff at all community sites, novel strategies to address barriers are needed.3 To address this challenge, we have explored telehealth strategies to conduct clinical research in community oncology practices in our network. This has led to the development of the Virtual Research Nurse (VRN) program, in which a research nurse located at LSU-LCMC Health Cancer Center in New Orleans can remotely screen, enroll, and follow patients on clinical trials at distant community oncology practices. Here, we outline the evolution of the VRN program and propose a framework for implementing VRNs in other community oncology settings.
Program Development
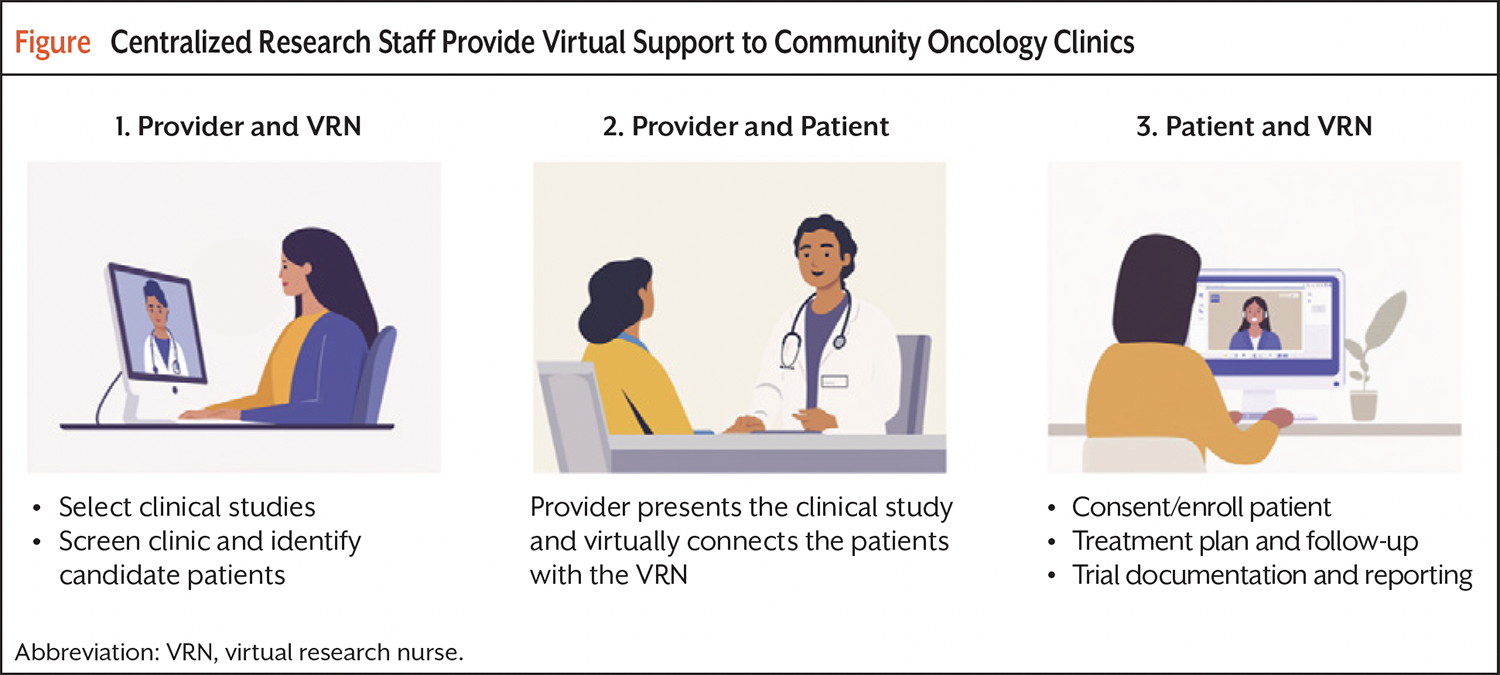
The VRN model is summarized in the Figure. Briefly, following the establishment of data user agreements, the VRN can remotely access the provider’s clinic schedule and patient medical records to identify potential trial candidates ahead of their scheduled appointment. The VRN informs the provider, who discusses the clinical trial with the patient. If the patient expresses interest, they are immediately connected with the VRN by teleconference, during which the VRN explains the protocol, answers patient questions, and obtains consent. The VRN works with the staff at the community practice to schedule protocol requirements and establish a treatment or follow-up plan and remains in contact with the patient through phone calls, electronic messaging, or virtual visits. All trial documentation and reporting are completed by the VRN.
Early use cases of a VRN in the Gulf South CTN have identified a number of best practices for the VRN model. The first use case had a VRN screen patients for clinical trials at a rural hospital without research staff. With a portfolio of 3 clinical trials, the VRN screened 170 patients at this site, but there were no accruals. From this effort, the Gulf South research team realized that community oncology providers need to have a diverse portfolio of trials to address different malignancies across the full continuum of cancer care and a provider “champion” willing to help overcome potential roadblocks.
Next, the Gulf South research team was interested in testing the ability to screen and manage trial patients remotely in a suburban oncology clinic with an established research program that included dedicated staff and a comprehensive trial portfolio for their patient population. We found that the enrollment rate for patients screened virtually was similar to patients screened on-site. This effort demonstrated the feasibility of a VRN as part of a well-established research program.
The lessons learned from the early uses of a VRN were essential with the arrival of the COVID-19 pandemic, which presented research challenges due to limited patient access for research staff and staffing shortages. To address these challenges, the Gulf South CTN used VRNs to screen and manage patients for clinical trials remotely as needed. Thus, VRNs represented an effective resource to adapt to changing environments in community oncology practices.
Through the initial development of the VRN framework in the Gulf South CTN, it was clear that research-trained personnel, in this case registered nurses, could easily screen and identify potential candidates for clinical studies in urban and rural clinics. But several key components would enable a successful VRN program. First, VRNs should be research nurses with significant experience enrolling patients in clinical trials and the ability to rapidly build trust with remote investigators and patients. Additionally, participating sites need motivated provider investigators to lead in the creation of a comprehensive clinical trial portfolio by selecting trials for their patients.
Program Outcomes and Evaluation
We have identified 4 outcomes by which to evaluate the VRN program: clinical trial enrollment in community oncology practices, demographic representation, patient experience, and structural empowerment among research staff. Clinical trial enrollment will be measured by the frequency of accruals at the community site. The program specifically targets community sites for which the lack of research resources poses a barrier to clinical trial enrollment. Thus, the VRN program will facilitate clinical trial participation in a patient population that would otherwise have minimal access to research.
While diversity in clinical trials may be improving, there is still inequity in trial participation and a need to increase enrollment among minorities and underserved patients. One way to address this is to ensure patients have access to trials in community oncology practices in minority and underserved areas, like those served by the Gulf South CTN. We hypothesize that patients in community oncology settings will reflect the diversity of their community. We will evaluate the demographics of patients screened through the VRN program compared with cancer registry populations to assess representation and access to clinical trials.
We acknowledge that the patient’s experience is an important dimension of measured success in healthcare, and patient feedback can inform improvements in healthcare delivery. To measure the experience of clinical trial participants in the VRN program, we will administer the Research Participant Perception Survey-Ultrashort (RPPS-U) to patients at least 6 months after enrolling into the trial.7 The development of the survey was based on 5 key dimensions of the patient experience in clinical trials: informed consent, trust, coordination of care, information and communication, and respect for participant preferences.8 The RPPS-U has 13 questions—5 on patient characteristics, 2 on overall experience, and 6 questions from the long version—that accounted for 96% of the overall rating score. The RPPS-U was shown to produce reliable results with a higher completion rate than longer versions of the survey.7
There are also perceived organizational benefits from having a centralized research nurse. One benefit is the ability to implement new evidence-based practices and emerging technology quickly and efficiently. The use of centralized nurses also enables comprehensive mentorship and training for research staff. For this reason, we believe that a potential outcome of the VRN will be structural empowerment among research staff. The concept of structural empowerment centers around the definition of power in an organization, defined as the ability to mobilize information, resources, and support.9 Structural empowerment measures the extent to which employees have access to power structures in the workplace and has been found to correlate with professional quality of life and affective commitment among nurses in various settings.10-12 To measure this outcome, we will administer the Conditions for Workplace Effectiveness Questionnaire to the research staff in the VRN program.13 This survey has 12 questions that measure 4 constructs: access to opportunity, information, support and resources, and construct validity has been established.13
Evaluation and Reporting
Under the VRN framework, there will be continuous communication between research staff and physicians/investigators. Evaluation and reporting will occur at weekly, quarterly, and annual intervals. VRN will meet with investigators weekly to review potential trial participants and discuss the trial portfolio.
Investigators and the research team will also meet quarterly at the community site to review a detailed report of screenings, accruals, and operational barriers. Both weekly and quarterly meetings will provide an opportunity for investigators to identify new opportunities to open additional trials.
Finally, the research team will prepare an annual report of screenings and accruals that includes cross-tabulation by trial types, cancer types, and patient demographics.
Conclusion and Future Directions
We have described the VRN program implemented at the Gulf South CTN and provided a framework for implementing and evaluating VRN programs in other community settings. This approach addresses a common barrier to clinical trial enrollment at community oncology practices, the lack of research infrastructure.
While the program was developed using research nurses, the VRN model can be implemented with clinical research coordinators. We do anticipate that there will be challenges that will need innovative solutions, for example, addressing the distribution of experimental drugs and collection of biospecimens in rural clinics. Nonetheless, we believe the VRN model provides a viable approach to increase access to various types of clinical trials at community oncology practices with diverse patient populations.
Funding
This work was supported in part by the National Institutes of Health (NIH), National Cancer Institute (NCI; UG1CA189854).
Acknowledgments
NIH/NCI did not play a role in the design of the study; the collection, analysis, and interpretation of the data; the writing of the manuscript; and the decision to submit the manuscript for publication.
References
- McCaskill-Stevens W, Lyss AP, Good M, et al. The NCI Community Oncology Research Program: what every clinician needs to know. Am Soc Clin Oncol Educ Book. 2013;33:e84-e89.
- Unger JM, Vaidya R, Hershman DL, et al. Systematic review and meta-analysis of the magnitude of structural, clinical, and physician and patient barriers to cancer clinical trial participation. J Natl Cancer Inst. 2019;111:245-255.
- Ebrahimi H, Megally S, Plotkin E, et al. Barriers to clinical trial implementation among community care centers. JAMA Netw Open. 2024;7:e248739.
- Tang C, Hess KR, Sanders D, et al. Modifying the clinical research infrastructure at a dedicated clinical trials unit: assessment of trial development, activation, and participant accrual. Clin Cancer Res. 2017;23:1407-1413.
- Ludmir EB, Adlakha EK, Chun SG, et al. Enhancing clinical trial enrollment at MD Anderson Cancer Center satellite community campuses. Acta Oncol. 2019;58:1135-1137.
- Porter M, Ramaswamy B, Beisler K, et al. A comprehensive program for the enhancement of accrual to clinical trials. Ann Surg Oncol. 2016;23:2146-2152.
- Kost RG, de Rosa JC. Impact of survey length and compensation on validity, reliability, and sample characteristics for Ultrashort-, Short-, and Long-Research Participant Perception Surveys. J Clin Transl Sci. 2018;2:31-37.
- Yessis JL, Kost RG, Lee LM, et al. Development of a research participants’ perception survey to improve clinical research. Clin Transl Sci. 2012;5:452-460.
- Kanter RM. Men and Women of the Corporation. 2nd ed. Basic Books; 1993.
- Ruth-Sahd LA, Grim R. Nurse educators: professional quality of life related to conditions of work effectiveness. Nurse Educ. 2021;46:E55-E59.
- Kelly C, Barattucci M, Ahmad MS. Job satisfaction as a mediator between structural empowerment and intent-to-leave: a study of critical care nurses. Intensive Crit Care Nurs. 2022;70:103194.
- Laschinger HK, Finegan J, Shamian J, Casier S. Organizational trust and empowerment in restructured healthcare settings. Effects on staff nurse commitment. J Nurs Adm. 2000;30:413-425.
- Laschinger HK, Finegan J, Shamian J, Wilk P. Impact of structural and psychological empowerment on job strain in nursing work settings: expanding Kanter’s model. J Nurs Adm. 2001;31:260-272.
Copyright © 2025 Journal of Oncology Navigation & Survivorship. Reprinted with permission.