Approximately 40% of American men and women will be diagnosed with cancer at some point in their lives,1 with the cost of cancer care in the United States projected to exceed $245 billion by 2030.2
Rising cancer treatment costs strain family budgets and healthcare plan funds, which directly impact patient care. For example, a novel biologic therapy may cost more than $10,000 a month, which is double the US median monthly household income.3 Biologic-based medicines accounted for 38% of US prescription drug spending in 2015 and 70% of drug spending growth between 2010 and 2015.4
A biosimilar is a biologic agent with no clinically meaningful difference from its FDA-approved reference product; hence, biosimilars are approved by the FDA via a truncated process. As part of the shortened approval process, key characteristics, including purity, molecular structure, and bioactivity, are evaluated against the reference product, and they are thus substantially less expensive to achieve FDA approval compared with the reference product.6
Although data are limited, one study estimated that by introducing biosimilars into clinical practice, the US health system could save $54 billion by 2026, with savings ranging between $25 billion and $150 billion.4 Expanding the biosimilar market, especially in oncology, allows clinicians to identify less costly medications to advance cancer treatment and supportive care.5
Rising Adoption of Biosimilars
Biosimilar drug use has increased in the United States. Between 2018 and 2023, the biosimilar industry grew by 12.5% annually and now accounts for more than 46% of spending.7
Moreover, launches and usage of biosimilars are expected to substantially increase overall spending by $20 billion to $49 billion by 2027. This increase in spending reflects how biosimilars are encroaching on the market share of the reference drugs.7
Clinician Education Key to Mitigating Slow Uptake of Biosimilars
Despite the potential to lower costs of cancer care with biosimilars, cost savings have been limited in the United States.8 Knowledge gaps surrounding safety and efficacy among clinicians and patients have resulted in slower uptake of biosimilars in the United States.
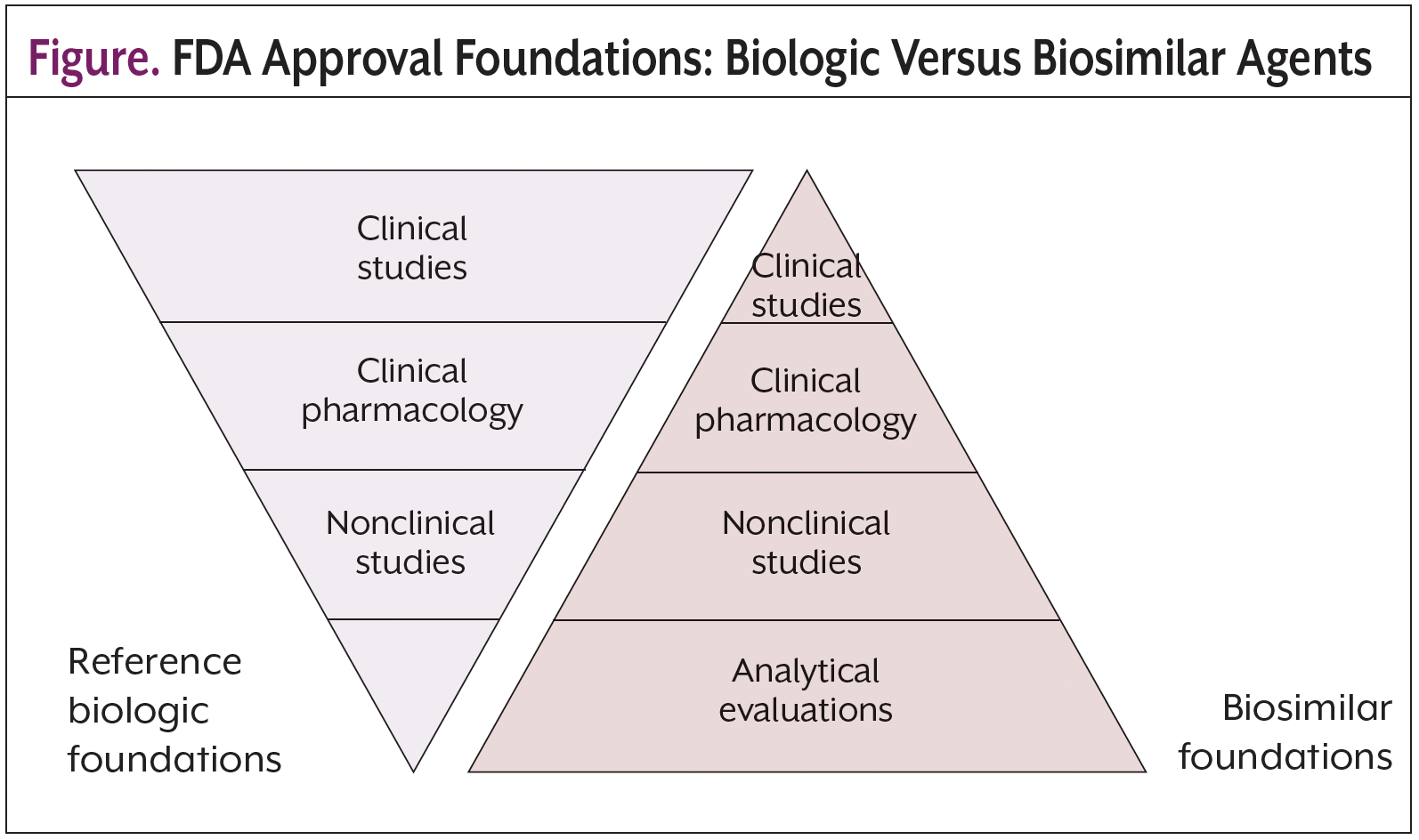
Educational programs are available to support clinician and patient understanding regarding biosimilar development, regulatory policies, and preclinical and clinical evidence supporting the efficacy and safety of biosimilars.8 One point of emphasis has been that all FDA-approved biologic agents undergo a rigorous evaluation, and biosimilar manufacturers use comparisons to the reference product to support approval and establish safety and effectiveness. This process is intended to avoid the expense and time commitments associated with clinical trials (Figure). Despite this truncated process, the pathway to biosimilar approval is still rigorous.8
Interchangeability
Unlike generic versus brand drugs, pharmacists cannot automatically substitute a biosimilar agent for the reference biologic agent without the prescriber’s permission. However, pharmacists may substitute a biosimilar drug for the reference biologic without permission if the FDA has determined that the biosimilar agent is “interchangeable.” For a biosimilar to receive a designation as interchangeable, additional clinical studies are required to demonstrate that the biosimilar agent would produce the same clinical result as the reference biologic in any given patient.9,10 This would include performing switching studies in which patients alternate between the reference biologic and the biosimilar multiple times over a specified period of time.8 Currently, no biosimilar drugs have been FDA-approved as interchangeable in oncology; however, the FDA has approved 4 interchangeable biosimilar products for certain ophthalmic and inflammatory diseases and diabetes.8
Notable Oncology Biosimilars
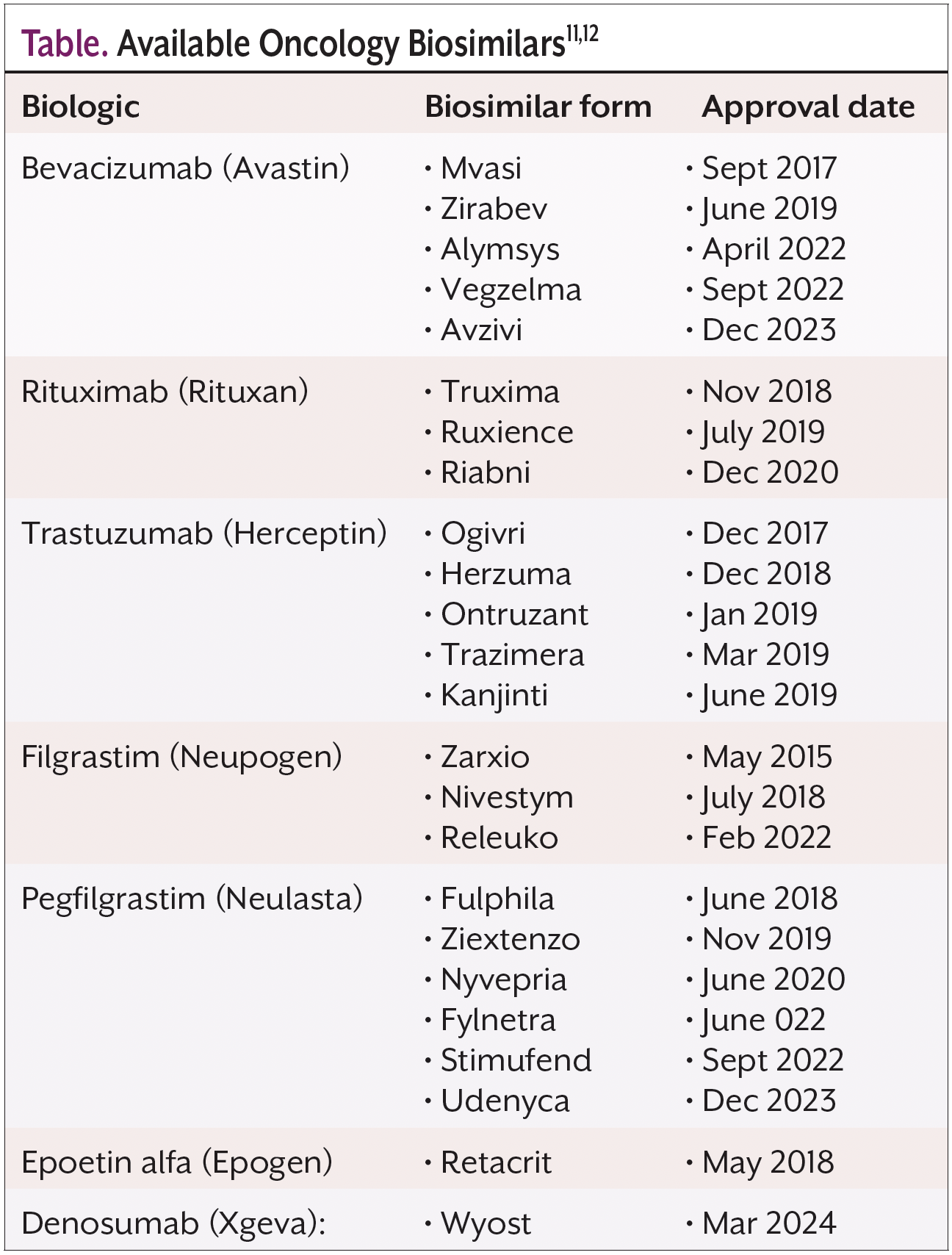
As of March 2024, the FDA has approved more than 20 biosimilars cancer treatments (Table)11,12 that work via various mechanisms of action.
Bevacizumab (Avastin) blocks vascular endothelial growth factor, a protein that some cancer cells produce in substantial amounts.13 Bevacizumab is used to treat colorectal; lung; glioblastoma; kidney; epithelial ovarian, fallopian tube, or primary peritoneal; cervical; and hepatocellular cancers.14 Biosimilars of bevacizumab include Mvasi, Zirabev, Alymys, Veglezma, and Avzivi. All biosimilars of bevacizumab are administered intravenously and are approved to treat the same cancers as bevacizumab, although they are not approved to treat liver cancer.15
Biosimilars can also treat cancer by targeting HER2, a protein that, when overexpressed, can cause cancer cells to grow out of control. Trastuzumab (Herceptin) binds to the extracellular domain of HER2, inhibiting its signaling.16 Biosimilars of trastuzumab include Ogivri, Herzuma, Kanjinti, Herzuma, and Ontruzant. Trastuzumab is approved to treat breast cancer and gastric or gastroesophageal junction adenocarcinoma, as are the trastuzumab biosimilars.17,18
PD-1/PD-L1 Inhibitors Biosimilars
Treatment with antibodies that inhibit the interaction between PD-L1 and its receptor, PD-1, can inhibit tumor growth.19
PD-1/PD-L1 inhibitors, such as pembrolizumab (Keytruda), are used as a front-line treatment for melanoma; non–small cell lung cancer; head and neck and squamous cell carcinomas; classical Hodgkin and primary mediastinal large B-cell lymphomas; urothelial, microsatellite instability-high or mismatch repair deficient, microsatellite instability-high or mismatch repair deficient colorectal, triple-negative breast, biliary tract, and gastric cancers; hepatocellular, Merkel cell, renal cell, and endometrial carcinomas; tumor mutational burden-high cancer; and cutaneous squamous-cell carcinoma.20
Biosimilar versions of pembrolizumab include InVivoSIM anti-human PD-1.21 SB27 is a biosimilar for pembrolizumab from Samsung Bioepis and is in phase 3 trials.22
The human immunoglobulin G1 kappa monoclonal antibody durvalumab (Imfinzi), used in adults with certain types of biliary tract cancers, also has a biosimilar product currently in development, InVivoSIM anti-human PD-L1 (durvalumab biosimilar).23
In addition, a biosimilar is under review for atezolizumab (Tecentriq), a monoclonal antibody used to treat urothelial carcinoma, non–small cell lung cancer, and small cell lung cancer, among other cancers.24
Looking Toward the Future of Biosimilars
Global cancer cases are expected to rise by 77% by 2050, reaching 35 million cases.25 As the use of oncology biosimilars increases, these affordable alternatives to the biologic reference drugs may lessen the financial impact on patients and payers. Pharmacists must stay current on the fast-moving biosimilar landscape as these new therapies come to market.
References
- National Cancer Institute. Cancer statistics. Updated September 25, 2020. Accessed April 22, 2024. www.cancer.gov/about-cancer/understanding/statistics
- American Hospital Association. The future of health system-based cancer. Accessed April 22, 2024. www.aha.org/sponsored-executive-dialogues/2023-02-28-/future-health-system-based-cancer-care.
- Liu Y. Utilizing oncology biosimilars to minimize the economic burden associated with cancer treatment: managed care considerations. Am J Manag Care. 2021;27(suppl 14):10.37765/ajmc.2021.88734.
- Mulcahy AW, Hlavka JP, Case SR. Biosimilar cost savings in the United States: initial experience and future potential. Rand Health Q. 2018;7(4):3
- Joshi D, Khursheed R, Gupta S, et al. Biosimilars in oncology: latest trends and regulatory status. Pharmaceutics. 2022;14(12):2721
- American Cancer Society. What are biosimilars? Updated April 27, 2022. Accessed April 22, 2024. www.cancer.org/cancer/managing-cancer/treatment-types/biosimilar-drugs/what-are-biosimilars
- IQVIA Institute. Biosimilars in the United States 2023-2027. January 31, 2023. Accessed April 22, 2024. www.iqvia.com/insights/the-iqvia-institute/reports-and-publications/reports/biosimilars-in-the-united-states-2023-2027
- Rodriguez G, Mancuso J, Lyman GH, et al. ASCO policy statement on biosimilar and interchangeable products in oncology. JCO Oncol Pract. 2023;19(7):411-419.
- Zlatkus A, Bixby T, Goyal K. Considerations for the US health-system pharmacist in a world of biosimilars. Drugs Context. 2020;9:2019-12-1.
- Overview of biologics and biosimilar products. American Pharmacists Association. Accessed April 22, 2024. www.pharmacist.com/Advocacy/Issues/Biosimilars
- List of biosimilars used in cancer treatment. American Cancer Society. Updated March 6, 2024. Accessed April 22, 2024. www.cancer.org/cancer/managing-cancer/treatment-types/biosimilar-drugs/list
- Food and Drug Administration. Biosimilar product information. Accessed April 23, 2024. www.fda.gov/drugs/biosimilars/biosimilar-product-information
- National Cancer Institute. Definition of bevacizumab. Accessed April 22, 2024. www.cancer.gov/publications/dictionaries/cancer-terms/def/bevacizumab
- Avastin (bevacizumab) injection, for intravenous use [prescribing information]. Genentech, Inc; Sept 2022. Accessed Aug 7, 2024. www.gene.com/download/pdf/avastin_prescribing.pdf
- Jacobsen S. The 4 biosimilars for Avastin, and what you should know about them. GoodRx Health. January 23, 2023. Accessed April 22, 2024. www.goodrx.com/avastin/biosimilar-drug
- Swain SM, Shastry M, Hamilton E. Targeting HER2-positive breast cancer: advances and future directions. Nat Rev Drug Discov. 2023;22(2):101-126.
- Herceptin (trastuzumab) intravenous infusion [prescribing information]. Genentech, Inc; June 2024. Accessed Aug 7, 2024. www.gene.com/download/pdf/herceptin_prescribing.pdf
- Jacobsen S. Trastuzumab biosimilars: what to know about Ogivri and other Herceptin alternatives. GoodRx Health. March 3, 2023. Accessed April 22, 2024. www.goodrx.com/trastuzumab/trastuzumab-biosimilars
- Abaza A, Sid Idris F, Anis Shaikh H, et al. Programmed cell death protein 1 (PD-1) and programmed cell death ligand 1 (PD-L1) immunotherapy: a promising breakthrough in cancer therapeutics. Cureus. 2023;15(9):e44582.
- Keytruda (pembrolizumab) injection, for intravenous use [prescribing information]. Merck; March 2024. Accessed Aug 7, 2024. www.accessdata.fda.gov/drugsatfda_docs/label/2024/125514s160lbl.pdf
- InVivoSIM anti-human PD-1 (pembrolizumab biosimilar). Bio X Cell. Accessed April 23, 2024. www.bioxcell.com/invivosim-anti-human-pd-1-pembrolizumab-biosimilar-sim0010
- Samsung Bioepis. Samsung Bioepis initiates phase 3 clinical trial for SB27, proposed biosimilar to Keytruda. Press release. Published April 5, 2025. Accessed December 4, 2024. www.samsungbioepis.com/en/newsroom/newsroomView.do?idx=385
- InVivoSIM anti-human PD-1 (durvalumab biosimilar). Bio X Cell. Accessed April 23, 2024. www.bioxcell.com/invivosim-anti-human-pd-l1-durvalumab-biosimilar-sim0027
- InVivoSIM anti-human PD-1 (atezolizumab biosimilar). Bio X Cell. Accessed April 23, 2024. www.bioxcell.com/invivosim-anti-human-pd-l1-atezolizumab-biosimilar-sim0009
- World Health Organization. Global cancer burden growing, amidst mounting need for services. February 1, 2024. Accessed April 24, 2024. www.who.int/news/item/01-02-2024-global-cancer-burden-growing--amidst-mounting-need-for-services