Gastric cancer ranks among the most prevalent cancers globally. A number of patients receive their diagnosis at advanced stages, primarily because the early symptoms are often subtle, and regular screening is infrequently conducted.1
Systemic chemotherapy is the mainstay treatment for metastatic gastric cancer; however, the use of targeted therapies has evolved significantly. Some examples of these therapies include ramucirumab, a monoclonal antibody that targets VEGFR2 protein; trastuzumab, a monoclonal antibody that targets HER2; and zolbetuximab, a monoclonal antibody that targets CLDN18.2.2
Ramucirumab was approved in April 2014 for the treatment of advanced or metastatic gastric or gastroesophageal junction (GEJ) adenocarcinoma with disease progression on or after prior fluoropyrimidine- or platinum-containing chemotherapy. Seven years later, trastuzumab deruxtecan was approved for HER2-positive gastric or GEJ adenocarcinoma in January 2021 for patients who have received a prior trastuzumab-based regimen.3 In October 2024, zolbetuximab was approved for patients with locally advanced unresectable or metastatic HER2-negative gastric or GEJ adenocarcinoma whose tumors are CLDN18.2 positive.4
Even with the approved targeted therapies for gastric cancer, many patients are still not receiving targeted therapy.
Data From AnswerY: A Real-World Patient-Physician Database
Real-world data can be used to further understand trends in biomarker testing and the use of targeted therapies among patients with gastric cancer. AnswerY, a proprietary database and platform owned by Amplity, the parent company of Oncology Practice Management, is a real-world database and platform built from HIPAA-compliant transcriptions of US prescriber–patient visits. Using AI and natural language processing (NLP), it extracts, visualizes, and summarizes treatment discussions and clinical decisions. Covering inpatient and outpatient care across 70+ specialties since 2017, AnswerY was known as Amplity Insights prior to January 2025.
AnswerY was searched for patients diagnosed with gastric cancer (from 2017 to 2025; grouped as before 2021 and after 2021) for any mention of “biomarker,” “biomarker tested,” and “biomarker positive.” It was also used to search for mentions of patients who were biomarker positive and prescribed any line of targeted therapy.
Before 2021, 76% of patients with gastric cancer were tested for biomarkers. After 2021, 69% of patients with gastric cancer were tested for biomarkers.
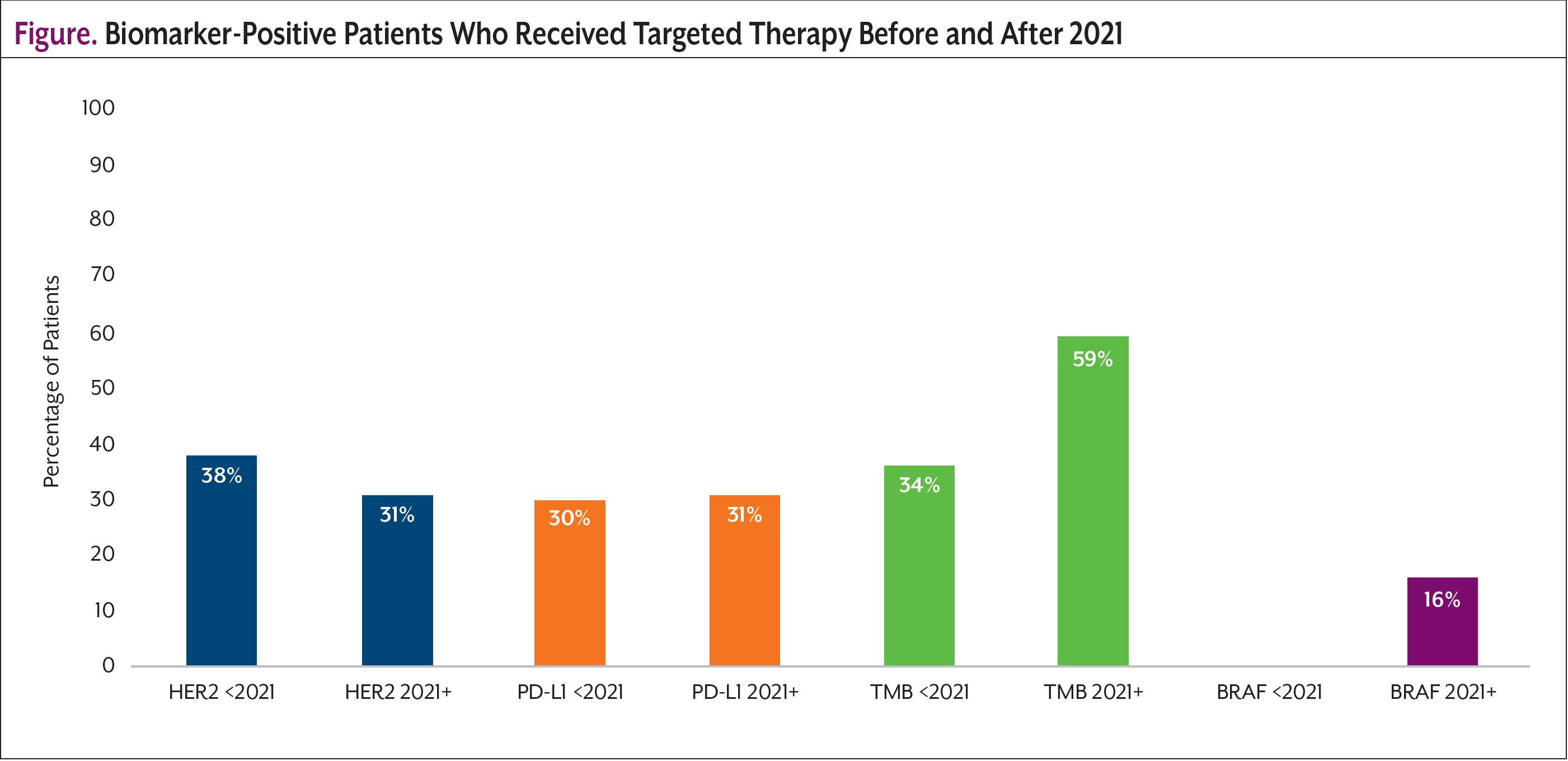
Among the patients who received biomarker testing, HER2 (<2021, 69%; 2021+, 65%) and PD-L1 (<2021, 25%; 2021+, 28%) biomarkers were found most frequently. The Figure shows the percentage of patients who were biomarker positive and received targeted therapy before and after 2021. Mentions for targeted therapy were found for 4 biomarkers: HER2, PD-L1, TMB, and BRAF. The number of patients who were biomarker positive and on a targeted therapy were low—between 30% and 38% before 2021 and 16% and 59% after 2021—with most patients on targeted therapy for HER2- and PD-L1–positive disease.
Low Rates of Biomarker-Positive Patients Receiving Targeted Therapy
This real-world analysis showed that not many patients who are biomarker positive are receiving targeted therapy. The low number of patients who were biomarker positive and on a targeted therapy may be due to multiple factors. One factor may be that chemotherapy is generally used in the first line of therapy, while targeted therapy is usually indicated in the second line of therapy,5,6 with the exception of zolbetuximab.7 Additionally, the use of targeted therapies is restricted to a specific subset of the population of patients with gastric cancer due to a small fraction of patients presenting with specific biomarkers.8
The low amount of targeted therapy being used in biomarker-positive patients may also be due to sociodemographic or socioeconomic factors. A study on sociodemographic disparities in targeted therapy for ovarian cancer included patients with ovarian cancer (stage I-IV) between 2012 and 2019 from the National Cancer Database.9 Among the 99,286 patients with ovarian cancer, only 4.1% received targeted therapy. Variations were observed in the administration of targeted therapies due to healthcare access, health insurance status, the age at which the diagnosis was made, the stage of the disease, and the presence of comorbidities at the time of diagnosis.
Conclusion
This analysis conducted in a real-world setting revealed that a limited number of biomarker-positive patients are being administered targeted therapy. This is consistent with what is seen in the literature and may be due to limited targeted therapies being used in the first-line setting as well as sociodemographic and socioeconomic factors. More research is needed to make targeted therapies available to patients with gastric cancer.
References
- Guan WL, He Y, Xu RH. Gastric cancer treatment: recent progress and future perspectives. J Hematol Oncol. 2023;16(1):57.
- American Cancer Society. Treating stomach cancer. Accessed April 7, 2025. www.cancer.org/cancer/types/stomach-cancer/treating.html
- FDA. FDA approves fam-trastuzumab deruxtecan-nxki for HER2-positive gastric adenocarcinomas [press release]. January 15, 2021. Accessed April 7, 2025. www.fda.gov/drugs/resources-information-approved-drugs/fda-approves-fam-trastuzumab-deruxtecan-nxki-her2-positive-gastric-adenocarcinomas
- FDA. FDA approves zolbetuximab-clzb with chemotherapy for gastric or gastroesophageal junction adenocarcinoma [press release]. October 18, 2024. Accessed April 7, 2025. www.fda.gov/drugs/resources-information-approved-drugs/fda-approves-zolbetuximab-clzb-chemotherapy-gastric-or-gastroesophageal-junction-adenocarcinoma#:~:text=gastroesophageal%20junction%20adenocarcinoma-,FDA%20approves%20zolbetuximab%2Dclzb%20with%20chemotherapy%20for%20gastric%20or%20gastroesophageal,%2C%20a%20claudin%2018.2%20(CLDN18
- Enhertu (fam-trastuzumab deruxtecan-nxki) for injection [prescribing information]. Daiichi Sankyo, Inc; January 2025.
- Cyramza (ramucirumab) injection, for intravenous use [prescribing information]. Eli Lilly and Company; April 2025.
- Vyloy (zolbetuximab-clzb) for injection, for intravenous use [prescribing information]. Astellas Pharma US, Inc; March 2025.
- Skórzewska M, Gęca K, Polkowski WP. A clinical viewpoint on the use of targeted therapy in advanced gastric cancer. Cancers (Basel). 2023;15(22).
- Amin SA, Collin LJ, Kavecansky J, et al. Sociodemographic disparities in targeted therapy in ovarian cancer in a national sample. Front Oncol. 2023;13:1104630.