Bispecific antibodies have emerged as a promising class of immunotherapies in oncology, offering innovative treatment options for various cancers. These engineered antibodies are designed to simultaneously bind 2 different targets, enhancing the immune system's ability to recognize and eliminate cancer cells.1 Notable examples include blinatumomab for acute lymphoblastic leukemia and teclistamab for multiple myeloma.2,3
Despite their therapeutic potential, the adoption and utilization of bispecific antibodies exhibit significant disparities, particularly between urban and rural healthcare settings.4 In urban areas, where advanced medical infrastructure and specialized oncology centers are more prevalent, the use of bispecific antibodies is more concentrated.4 Conversely, rural regions often face challenges in accessing these therapies, leading to a concentration of their use in cities.4
Real-world data can be used to further understand trends in the use of bispecific antibodies and to better recognize where there may be areas of inequity.
Challenges in Rural Access to Bispecific Antibodies
Several factors contribute to the limited use of bispecific antibodies in rural areas. First, infrastructure limitations play a significant role, as administering bispecific antibodies requires specialized medical expertise and organization, including inpatient oncology services capable of monitoring patients during the initial dosing phases.4 Many rural clinics lack the necessary facilities and trained personnel to safely administer these therapies.4 Financial constraints also pose a major barrier, as the high cost of bispecific antibody treatments creates a significant financial burden for rural healthcare facilities, which may lack the resources to invest in these advanced therapies.5 This financial strain further limits the availability of such treatments in rural settings.5 Additionally, the geographic distribution of clinical trials for these therapies exacerbates access issues.6 A study found that 34% of US states had no open clinical trials for chimeric antigen receptor T cells and bispecific antibodies.6 States with higher percentages of Black residents, who face a higher incidence of multiple myeloma, were particularly affected, suggesting that the current distribution of clinical trials contributes to disparities in access to advanced therapies.6
Data from a Real-World Patient-Physician Database
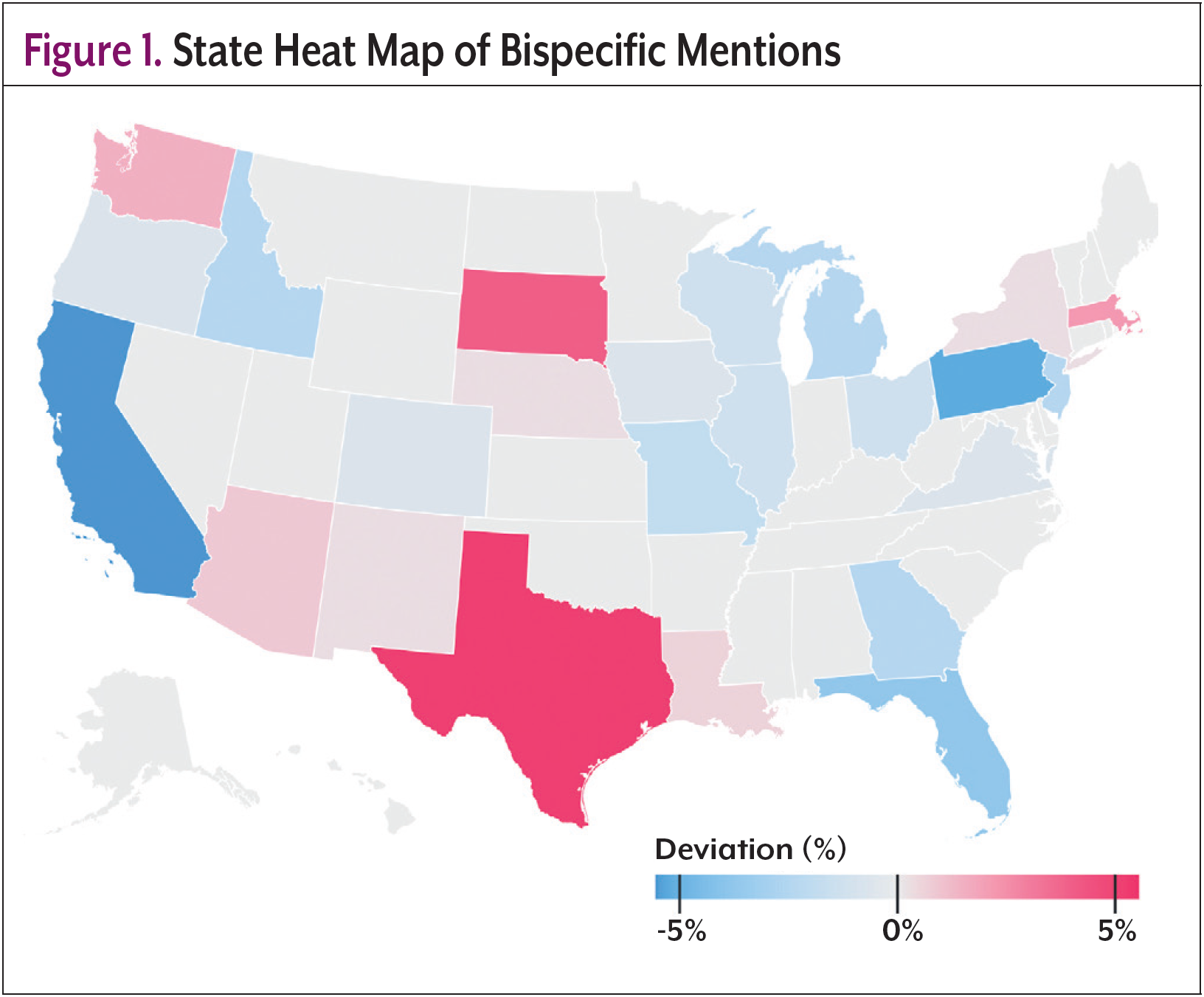
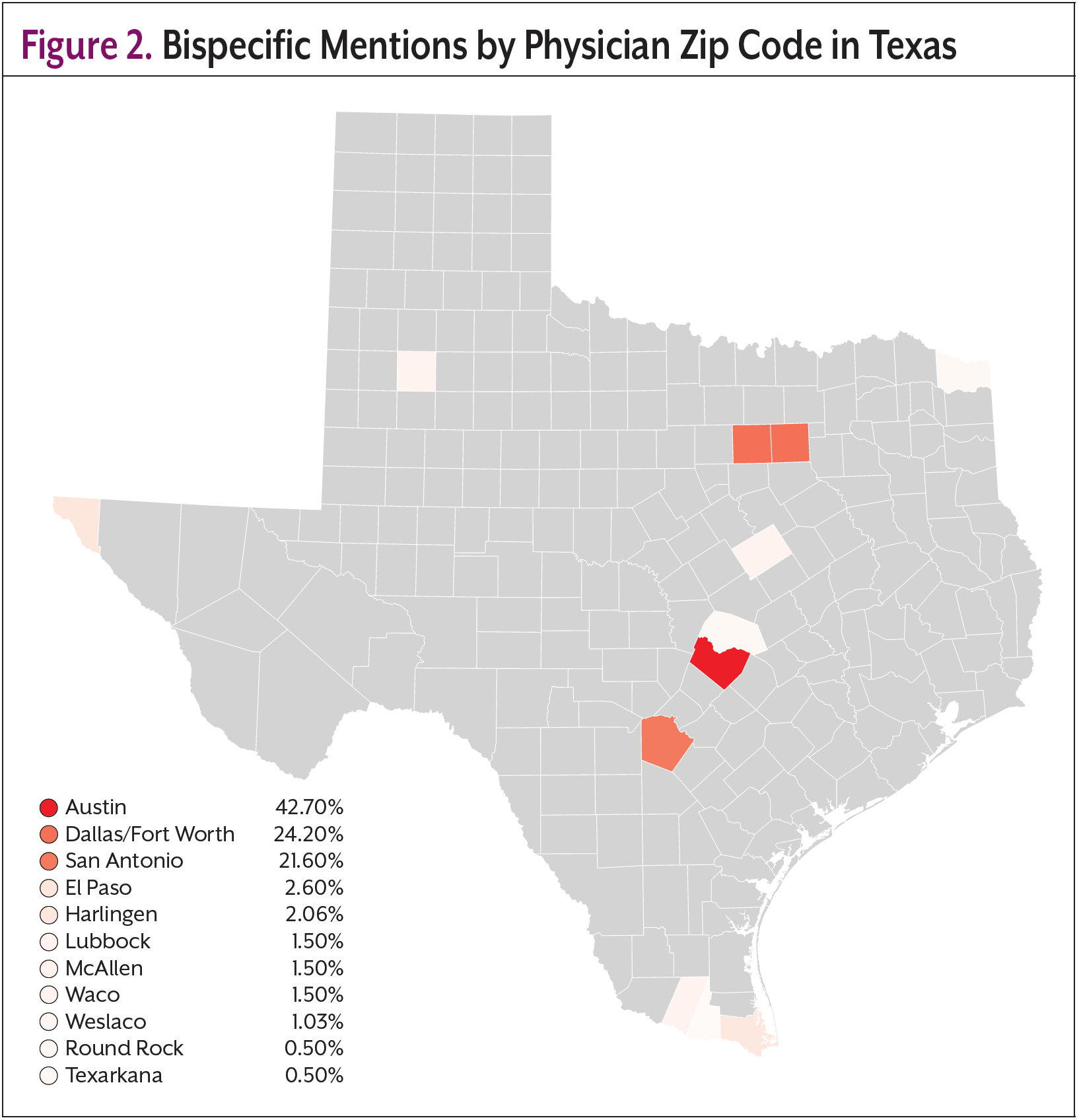
Real-world data can be used to further understand trends in the use of bispecific antibodies and to better recognize where there may be areas of inequity. AnswerY, a proprietary database and platform owned by Amplity, the parent company of Oncology Practice Management, is a real-world database and platform built from HIPAA-compliant transcriptions of US prescriber–patient visits. Using AI and natural language processing (NLP), it extracts, visualizes, and summarizes treatment discussions and clinical decisions. Covering inpatient and outpatient care across 70+ specialties since 2017, AnswerY was known as Amplity Insights prior to January 2025. AnswerY was searched for any mention of “bispecific antibody” across any indication. 864 patients matching 1819 records met these criteria. Figure 1 shows the geographic distribution of bispecific mentions in the United States, with red states demonstrating higher counts than would be expected compared with the overall database. An analysis of Texas revealed that most patients with records mentioning “bispecific antibodies” were treated by physicians in the zip codes of larger cities; 42.7% Austin, 24.2% Dallas/Fort Worth, and 21.6% San Antonio (Figure 2). Although there were some results in more rural areas of Texas, the raw records, in some cases, provided further insight into why this was the case. For example, when looking into the record of a patient-physician visit in Texarkana, it was found that this physician was a pain specialist and was seeing a patient who was on a bispecific antibody; however, this particular physician was not their treating oncologist. This may suggest that, although there was 1 record in Texarkana, this patient may still be traveling far distances to receive treatment. Findings from this real-world database support the need for increased efforts to increase adoption and utilization of bispecific antibodies in rural areas.
Efforts to Address Disparities
Recognizing these challenges, several initiatives are working to improve the accessibility of bispecific antibody therapies in rural areas. One such initiative has been spearheaded by the Association of Community Cancer Centers, which launched educational programs to prepare cancer care teams for the successful incorporation of these therapies into their practice.7 Additionally, organizations are sharing operational insights to assist in the delivery of bispecific antibodies, equipping care teams in community and rural cancer programs with the knowledge needed to administer these treatments effectively.4 Collaboration between academic centers and community practices is also crucial to improve access to bispecific antibodies. With a successful partnership, community oncologists can refer patients to academic centers for their initial treatment with the following cycles being managed by their local practices, while helping mitigate costs for community centers.4,5 Moreover, pharmaceutical companies and patient advocacy groups are offering financial support resources to help alleviate the financial burden on rural healthcare facilities, ensuring the availability of bispecific antibody therapies in community clinics and oncology centers.5
Conclusion
While bispecific antibodies represent a significant advancement in cancer treatment, their utilization is currently more concentrated in urban areas due to infrastructure, financial, and geographic challenges, which was supported by real-world data from the AnswerY database. Addressing these disparities is crucial to ensure equitable access to these therapies for all patients, regardless of geographic location. Ongoing efforts to integrate bispecific antibodies into community oncology practices and provide financial support are steps in the right direction toward eliminating these disparities.
About the Author

Abbey Nakano, PharmD, is an Associate Medical Director at Amplity and medical communications professional whose expertise involves translating medical information into engaging content.

John Perez, MBA, MSDS is the Director of Data and Analytics at Amplity Intel and specializes in AI-driven analysis of unstructured medical data for pharmaceutical insights.
References
- Wei J, Yang Y, Wang G, Liu M. Current landscape and future directions of bispecific antibodies in cancer immunotherapy. Front Immunol. 2022;13:1035276.
- Gökbuget N, Dombret H, Bonifacio M, et al. Blinatumomab for minimal residual disease in adults with B-cell precursor acute lymphoblastic leukemia. Blood. 2018;131:1522-1531.
- Usmani SZ, Garfall AL, van de Donk NWCJ, et al. Teclistamab, a B-cell maturation antigen × CD3 bispecific antibody, in patients with relapsed or refractory multiple myeloma (MajesTEC-1): a multicentre, open-label, single-arm, phase 1 study. Lancet. 2021;398:665-674.
- Braun A, Gouni S, Pulles A, et al. Bispecific antibody use in patients with lymphoma and multiple myeloma. Am Soc Clin Oncol Educ Book. 2024;44:e433516.
- Where to Start? Implementing Bispecific Antibody Therapies in the Community Setting. Accessed February 17, 2025. https://www.accc-cancer.org/acccbuzz/blog-post-template/accc-buzz/2024/12/18/where-to-start-implementing-bispecific-antibody-therapies-in-the-community-setting
- Alqazaqi R, Schinke C, Thanendrarajan S, et al. Geographic and racialdisparities in access to chimeric antigen receptor T cells and bispecific antibodies trials for multiple myeloma. JAMA Netw Open. 2022;5:e2228877.
- Successful Integration of Bispecific Antibodies into Community Oncology Practice. Accessed February 20, 2025. https://www.accc-cancer.org/home/learn/precision-medicine/treatment/bispecific-antibodies/successful-integration-of-bispecific-antibodies-into-community-oncology-practice?utm_source=chatgpt.com